Chapter 02 Protein Structure
Introduction
Protein structure was first identified by biochemist John Kendrew using the X-ray diffraction method on myoglobin at a 2-Å resolution.
- Primary Structure
The first level of hierarchy is the sequence of amino acids forming the initial polypeptide chain.
- Secondary Structure
Certain amino acids tend to form loops and simple shapes such as helices and sheets.
- Tertiary Structure (Native Structure)
The overall chain further folds into a compact, three-dimensional tertiary structure, constituting the third level of the hierarchy. This is the most stable structure as it optimizes the various attraction forces among the different amino acids in the chain.
These types of shapes are common, but some proteins, like fibrous proteins, have an elongated form. This form includes an arrangement of secondary elements but lacks the characteristic complex three-dimensional fold.
:max_bytes(150000):strip_icc():format(webp)/protein-structure-373563_final11-5c81967f46e0fb00012c667d.png)
Coenzymes and Prosthetic Groups
The amino acid chain is a central component but not necessarily the only component. Proteins may include other atoms or small molecules required for their function and stability.
These cofactors are chemically diverse and can be organic molecules or elements, either metallic (e.g., zinc) or non-metallic (e.g., selenium).
Enzymes, for example, often require cofactors to carry out their functions.
The common coenzymes NADH and FADH2 are well-known examples. These molecules, derived from the B-complex vitamins niacin (B3) and riboflavin (B2), serve as carriers and donors of high-energy electrons in reduction-oxidation (redox) reactions.
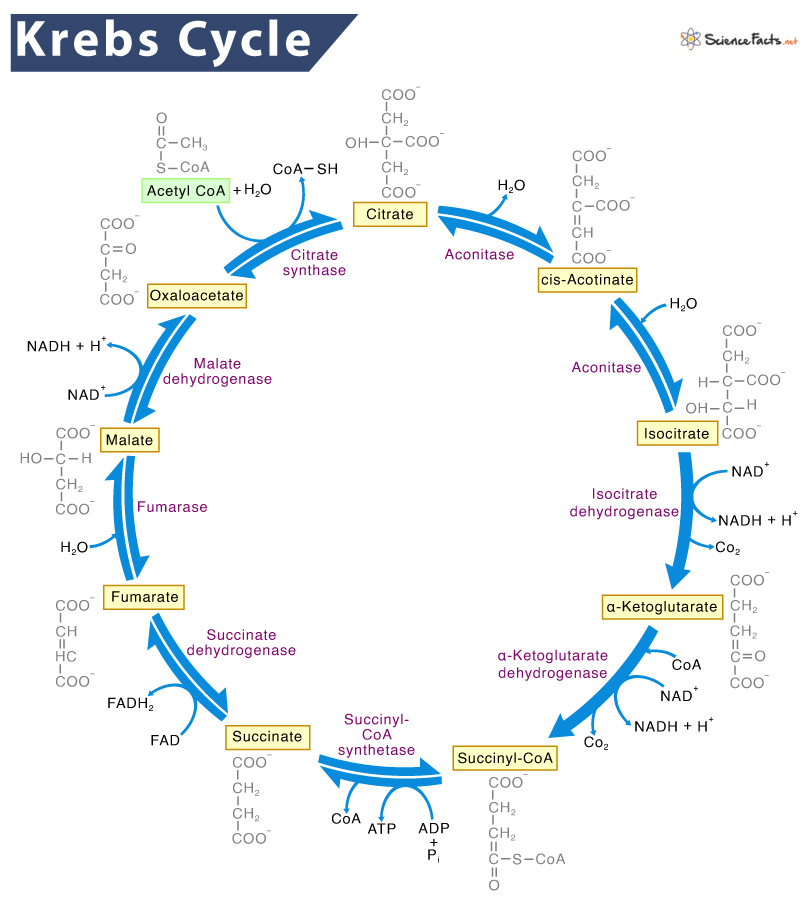
The Krebs cycle, also known as the citric acid cycle or the tricarboxylic acid (TCA) cycle, is a crucial metabolic pathway in cellular respiration. Here’s an explanation of its purpose and importance:
Purpose of the Krebs Cycle
The primary purpose of the Krebs cycle is to generate high-energy electron carriers that will be used in the electron transport chain to produce ATP, the energy currency of the cell. Here's how it works:
- Acetyl-CoA Production: The cycle begins with acetyl-CoA, a molecule derived from carbohydrates, fats, and proteins.
- Energy Carrier Production: As acetyl-CoA enters the cycle, it undergoes a series of chemical reactions that produce electron carriers, NADH and FADH2.
- ATP Production: These electron carriers then transfer their electrons to the electron transport chain, leading to the production of ATP.
Importance of the Krebs Cycle
-
Energy Production: The Krebs cycle is a key component of cellular respiration, which is the process by which cells produce energy. The cycle itself generates only a small amount of ATP directly, but it produces NADH and FADH2, which are essential for the production of a much larger amount of ATP in the electron transport chain.
-
Carbon Dioxide Production: The cycle produces carbon dioxide as a byproduct, which is then exhaled by organisms. This process helps to regulate the levels of carbon dioxide in the blood.
-
Intermediate Compounds: The Krebs cycle produces intermediate compounds that are used in various other biochemical pathways. For example, it provides intermediates for amino acid synthesis and for the production of other important molecules.
-
Metabolic Hub: The cycle is a central hub in cellular metabolism. It integrates the metabolism of carbohydrates, fats, and proteins, enabling the cell to efficiently produce energy from a variety of sources.
Steps of the Krebs Cycle
- Formation of Citrate: Acetyl-CoA combines with oxaloacetate to form citrate.
- Conversion to Isocitrate: Citrate is rearranged to form isocitrate.
- Oxidation to α-Ketoglutarate: Isocitrate is oxidized to α-ketoglutarate, producing NADH and releasing CO₂.
- Conversion to Succinyl-CoA: α-Ketoglutarate is further oxidized to succinyl-CoA, producing another NADH and releasing another CO₂.
- Formation of Succinate: Succinyl-CoA is converted to succinate, producing ATP (or GTP).
- Oxidation to Fumarate: Succinate is oxidized to fumarate, producing FADH2.
- Conversion to Malate: Fumarate is converted to malate.
- Regeneration of Oxaloacetate: Malate is oxidized to oxaloacetate, producing another NADH.
Each turn of the cycle processes one molecule of acetyl-CoA, and for each acetyl-CoA, the cycle produces:
- 3 NADH
- 1 FADH2
- 1 ATP (or GTP)
- 2 CO₂
These high-energy electron carriers (NADH and FADH2) then donate electrons to the electron transport chain, driving the production of a large amount of ATP through oxidative phosphorylation.
In summary, the Krebs cycle is fundamental for energy production, carbon dioxide removal, and providing key intermediates for various metabolic pathways, making it essential for the proper functioning of cells and organisms.
Redox Reactions
Some enzymes use several different cofactors. For example, pyruvate dehydrogenase (PDH) is a key enzyme in carbohydrate catabolism. The catabolic metabolism of carbohydrates begins with glycolysis, a 10-step biochemical pathway that turns one glucose molecule into two molecules of pyruvate, and continues with the citric acid (Krebs) cycle, which further degrades and oxidizes the remnants of pyruvate to CO₂.
PDH operates between these two major pathways, activating pyruvate so it can enter the Krebs cycle. Specifically, PDH catalyzes the oxidative decarboxylation of pyruvate to acetyl-CoA (ACoA), its activated form.
PDH is not a single enzyme but a three-component complex, with each component participating in a different step of pyruvate activation, each using a different coenzyme:
-
Thiamine Pyrophosphate (TPP): A derivative of thiamine (vitamin B1), TPP enables the first component of PDH to decarboxylate pyruvate into acetaldehyde, releasing CO₂. A deficiency in thiamine can lead to beriberi, which affects several body systems.
-
Lipoic Acid: This prosthetic group is covalently attached to the enzyme. It catalyzes the oxidation of the substrate’s hydroxyl-ethyl group and its transfer to coenzyme A (CoA), yielding acetyl-CoA. CoA is derived from pantothenic acid (vitamin B5). The process involves the reduction of the disulfide bond in lipoic acid to two thiol groups, coupling the oxidation of the hydroxyl-ethyl group to acetyl-CoA.
-
Flavin Adenine Dinucleotide (FAD): The third component of PDH uses FAD to re-oxidize the thiol groups in lipoic acid, restoring the disulfide bond. FAD itself is reconstituted by NAD⁺, which is reduced to NADH.
Interestingly, the PDH reaction is also used by α-ketoglutarate dehydrogenase, an enzyme in the Krebs cycle, but with a different substrate (α-ketoglutarate instead of pyruvate). The reuse of successful protein functions is a key evolutionary strategy.
-
Glycoproteins: Proteins that bind a large number of sugar moieties (glycans).
-
Lipoproteins: Proteins that bind lipid groups, forming large spherical fatty bodies. They are used for the transport of fatty acids and cholesterol between different organs.
-
Hemoproteins: Proteins that contain an organic group called ‘heme’, which includes an iron atom.
- Hemoglobin: Transports oxygen and carbon dioxide between the lungs and peripheral tissues.
- Myoglobin: Serves as a temporary reservoir of oxygen in muscles.
- In both hemoglobin and myoglobin, the heme group functions in binding oxygen, and is essential for protein function.
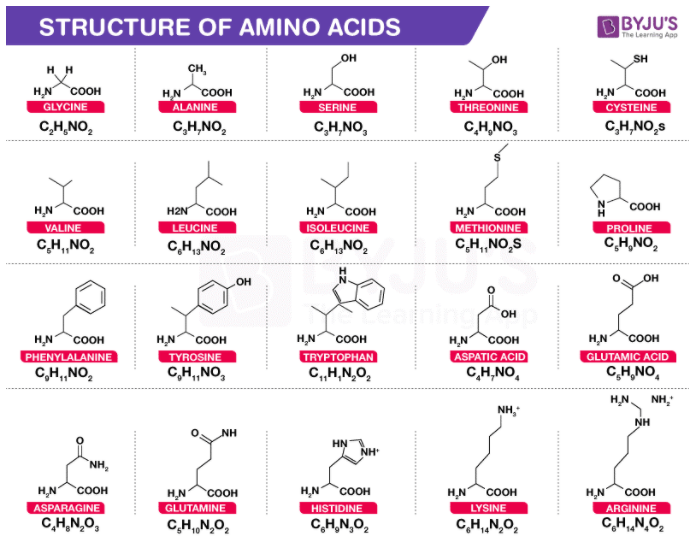
Primary Structure
The exact sequence of the protein is very important, as it determines the final fold, and therefore the function, of the protein.
-
amino acids and their physicochemical properties
-
how the amino acids connect to each other
-
Central Carbon Atom (Cα):
- Each amino acid has a central carbon atom called the ‘α-carbon’ or ‘Cα’.
- The Cα is surrounded by four substituents:
- A hydrogen atom
- An amino group (‘α-amino’)
- A carboxyl group (‘α-carboxyl’)
- A side chain (unique to each amino acid)
-
α-Carboxyl Group:
- Low pKa (~2)
- Tends to be deprotonated and negatively charged at physiological pH (~7)
-
α-Amino Group:
- High pKa (9–10)
- Tends to be protonated and positively charged at physiological pH
-
Charges in Protein Chains:
- Charges of the α-amino and α-carboxyl groups are nullified when amino acids are integrated within the protein chain.
- Charges remain at the two ends of the protein sequence.
-
Backbone vs. Side Chain:
- Backbone:
- Includes all non-side chain atoms (Cα, its hydrogen atom, the α-carboxyl group, and the α-amino group).
- Identical across all amino acids.
- Side Chain:
- Different for each amino acid.
- Determines the unique properties of each amino acid.
- Amino acids are often referred to as ‘residues’ when incorporated within the protein chain.
- Backbone:
-
Configuration of Amino Acids:
- Affects protein structure and function.
- Important property, though unrelated to the specific nature of the amino acid side chain.
Equation of PKA